Attribution error results from a positive stereotype
A 58-year-old man with type 1 diabetes at age 38, a case of latent autoimmune diabetes of adulthood, reports worsening control of his blood sugars despite increasing doses of insulin. An internist must sort through the facts of the case to find out what's responsible.
Lynn Bentson, FACP, an internist in Albany, Ore., told us about the case of a 58-year-old man with diabetes mellitus and increasing insulin requirements. The patient was diagnosed with type 1 diabetes at age 38, a case of latent autoimmune diabetes of adulthood.
Dr. Bentson has a special interest in diabetes and cares for many of the type 1 diabetics in her town, including managing preexisting diabetes in pregnant women and patients on insulin pumps. This patient was referred from a free clinic in a neighboring community for help with diabetes management.
The patient was self-employed as a house painter, and was very proactive about his health. He ran several times a week, played basketball regularly, attended yoga classes, and ate only organic foods. Because of his diabetes, he was unable to purchase affordable health insurance, but he obtained a glucose monitor and test strips from a free clinic. He checked his blood glucose meticulously, at least four times a day, and graphed the results monthly on his computer to track his glucose control. For many years his diabetes was well controlled, with hemoglobin A1c measurements between 6.8% and 7.3%.
Dr. Bentson first saw the patient in July 2009 because of worsening control of his blood sugars despite increasing doses of insulin. At that time, his total daily insulin dose was 80 units: 70 units of insulin aspart and 10 units of insulin glargine. On physical examination, the patient was an intelligent, muscular man who appeared well. He weighed 89 kg with a BMI of 26. He had no retinopathy or neuropathy. His hemoglobin A1c was 8.5%. The patient brought in detailed records showing that over the prior month, he had significant hypoglycemia with blood glucose less than 50 mg/dL on five occasions, glucose of greater than 300 mg/dL on seven occasions, and 31 glucose readings between 200 and 300 mg/dL.
Dr. Bentson encouraged the patient to alter his regimen so that approximately half of his total daily insulin dose was basal insulin. She loaned him her favorite book on insulin therapy, and was able to get a “scholarship” for him to see a local diabetes educator with more than 20 years of experience. Despite the change in his insulin regimen and the consultation with the educator, his blood glucose control did not improve.
Dr. Bentson wondered if the patient might be having nocturnal hypoglycemia. She instructed him to wake up at 2 a.m. to check his blood sugar, but there was no evidence of nighttime falls in blood glucose.
Dr. Bentson told us that the patient's weight had been stable and there was no change in his diet or exercise. “I began to think about causes of insulin resistance,” she said. She considered concurrent endocrine disorders, including hyperthyroidism, acromegaly, pheochromocytoma and Cushing's syndrome. She also considered celiac disease. He did not have any clinical features of these conditions, and laboratory testing was negative.
“I wondered if he was not actually insulin deficient, but C peptide was undetectable,” she told us. “No insulin antibodies either.” She arranged for the patient to wear a three-day glucose monitor. “This did not show an exaggerated dawn phenomenon, and confirmed that it took a lot of insulin to make his sugars budge.” He was up to 100 units of insulin daily with even worse control of his blood sugars. “I asked him to stop all the supplements that had been prescribed by a naturopath, but this had no effect on his sugars,” she said.
“At this point, he had an on-the-job injury and was temporarily eligible for the Oregon Health Plan,” Dr. Bentson told us. “So I arranged for him to see an endocrinologist at the university.” Additional lab work was obtained, including serum protein electrophoresis, insulin-like growth factor 1 and 24-hour urine free cortisol, all of which were normal. His insulin was switched to a different type but “nothing changed.” His insulin dose increased further to 110 units per day with no improvement in glucose control.
Two further consultations, one with a second diabetes educator and another with a different endocrinologist, did not reveal a reason for the patient's poor control of blood glucose and escalating insulin needs.
“I saw him again in January 2011. At that visit, the patient asked me if there were ever problems with the injection site, because he had read about this in the book that I had loaned him earlier,” Dr. Bentson told us.
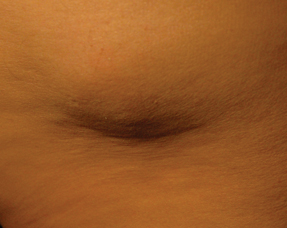
She replied that lipodystrophy was once a frequent problem, particularly with the use of animal insulins. But this patient had never used animal insulin. The patient went on, “I have been injecting the same site for the past 20 years and it feels hard.”
Dr. Bentson had the patient show her the site on his flank. “It felt as hard as a table top,” she told us.
Dr. Bentson and the patient agreed to change his injection site to the abdomen. “And from that day forward, his insulin resistance was cured,” Dr. Bentson said. His insulin requirements fell almost immediately from 110 units to 30 units a day with a subsequent drop in his hemoglobin A1c to 7% two months later.
In retrospect, Dr. Bentson told us that she had questioned the patient about his injection site, asking specifically whether he was injecting insulin into his arms because in his work as a house painter this might have affected insulin absorption. She realized now that this was a close-ended question, rather than the open-ended question “Where do you inject your insulin?” Despite evaluation by an experienced internist and consultations with two endocrinologists and two different diabetes educators, the issue of the injection site was not further explored.
It appears that the primary cognitive bias here was an attribution error. “This patient was very knowledgeable,” Dr. Bentson observed, “and seemed to know everything about his diabetes. Since he knew about insulin-to-carbohydrate ratios, I assumed that he would know about rotating his injection site.”
More commonly, doctors make attribution errors when patients fit a negative stereotype. For example, when a patient is unshaven and in shabby clothes with an enlarged liver, it is assumed that he is an alcoholic. In this case the stereotype was a positive one, that of an informed, proactive patient who was doing everything “right.”
“I was looking for something exotic and unusual,” Dr. Bentson observed. “It ended up being very basic.”



